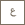Harnessing nanoparticles for COVID testing

Testing is vital for monitoring and mitigating the spread of SARS-CoV-2, the virus that causes the disease known as COVID-19. Further, community testing is integral to reopening and allowing a resumption of the daily patterns of life. KAUST Assistant Professor of Bioscience, Mo Li and his colleagues in the KAUST R3T think they may have a solution.
Tests approved by the World Health Organization (WHO), the US Centers for Disease Control (US CDC) and the Saudi Center for Disease Control (SCDC), start with extraction of genetic materials, which include the genome of the virus if present, from a nasal swab. Because of the potential presence of viruses, this step must be handled with extreme care. This step, therefore, requires a high level of biosafety containment and special reagents to be carried out successfully.
As the pandemic progresses, both the resources and commercial reagents needed for sample extraction dwindle relative to the burgeoning case numbers. Li and colleagues have recognized this challenge as a critical hurdle to improving testing availability.
A two-pronged approach
The first step is lowering the biosafety risks, so that the extraction step may be carried out in normal academic labs. KAUST Professor of Bioscience Arnab Pain and his research group have shown that the swab samples can be completely inactivated by a harsh chemical cocktail.
The samples can be safely handled in academic labs, and the viral genome may be extracted for follow-up tests using commercial kits. However, not only are these commercial kits in short supply, they are also difficult to automate due to centrifugation steps.
KAUST Assistant Professor of Bioscience Mo Li developed a graphic to explain how his team is approaching the streamlining of COVID-19 diagnostic testing. Graphic courtesy of Mo Li.
View full image
The second step is developing an in-house extraction kit that is compatible with inactivated samples to alleviate the shortage of commercial kits, as well as to enable automated sample preparation. Superparamagnetic nanoparticles (MNPs) can be used to isolate genetic materials such as the SARS-CoV-2 genome. These MNPs can be conveniently collected with a magnet without centrifugation, making them ideal for automation.
Unfortunately, the reagent we used to inactivate the viruses also inactivated commercial MNPs. Luckily, KAUST Associate Professor of Bioscience Samir Hamdan identified an alternative protocol for making silica-coated MNPs. Li found evidence that silica MNPs may survive the inactivation regents. He then synthesized silica MNPs from a few chemicals that are in abundant supply.
Preliminary success
"I hope to optimize the MNP protocol further and test them in an automatic liquid-handling robotic platform. If successful, this technology will make it possible to test more people and to process hundreds of samples in parallel in a timely manner," Li said.
These in-house MNPs have been tested in 24 inactivated COVID-19 swab samples by Li's lab and Pain's lab. The preliminary results show that the in-house MNP-based protocol successfully extracted viral genomes that are detected by the US CDC approved COVID-19 tests. The latest optimized MNP protocol developed in Li's lab outperformed the commercial product 75% of the time.
Li's current pilot batch of synthesis is estimated to be enough for around 10,000 samples. The production is straightforward to scale up. The beauty of these MNPs is that they are easy to use, stable at room temperature, and potentially amenable to automated high-throughput sample preparation.
"I want to thank KAUST Professors Niveen Khashab and Jean-Marie Basset for generously sharing their materials and knowledge," Li said.
Related stories
- How antibody testing can stem the spread of COVID-19
- Shining a light on the SARS-CoV-2 virus
-
Serving the Kingdom's COVID-19 research