KAUST stem cell research improves understanding of early human development

- Improved stem cell-derived embryo models allow scientists to study these processes more reliably in the lab
This study provides a new platform for research in reproductive medicine
Researchers at King Abdullah University of Science and Technology (KAUST) have developed a new method to improve how stem cell-derived embryo models are formed and controlled. The study, led by Associate Professor Mo Li, within KAUST's Division of Biomedical Sciences, provides novel insights into the earliest stages of human development.
These earliest stages of human development are fundamental to reproductive health and disruptions during this window can lead to infertility, early pregnancy loss, or developmental disorders. Despite decades of research, the mechanism that initiates and drives human embryo formation has not been fully understood.
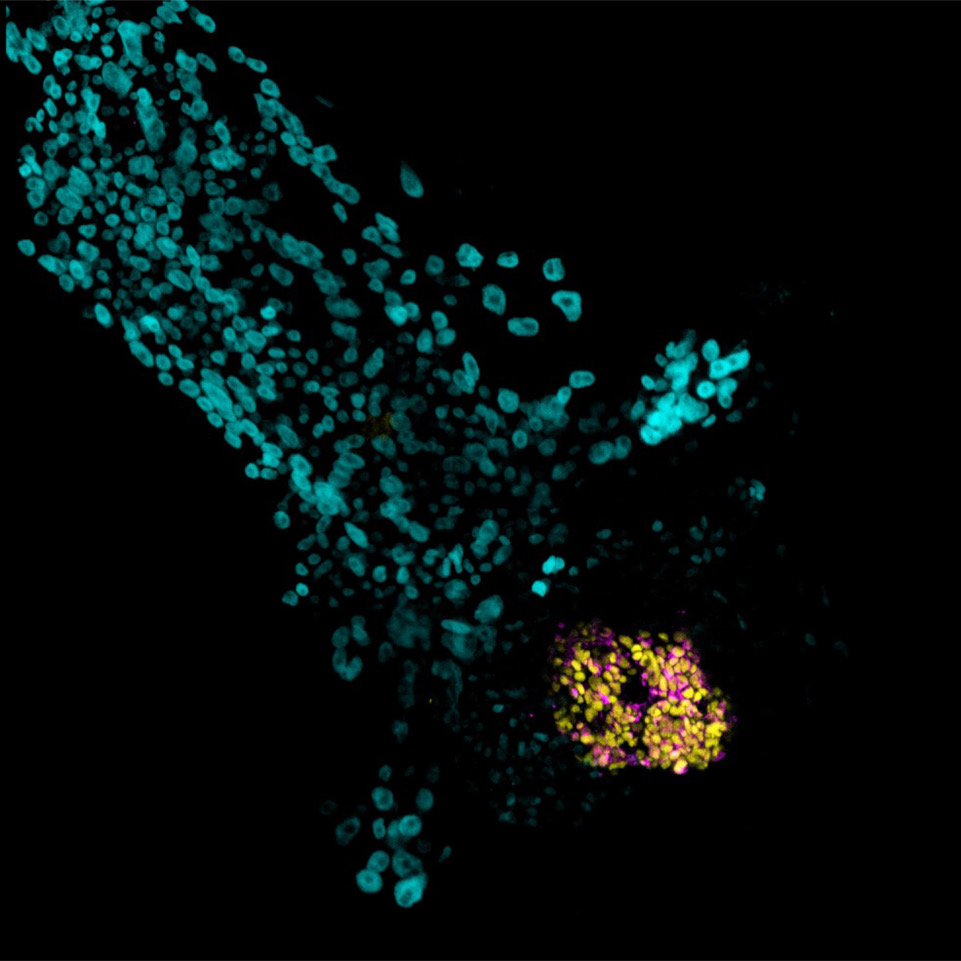
To study this process, the research team used human stem cell-derived blastoids —laboratory models that replicate key features of early human development— to observe otherwise inaccessible processes in a controlled laboratory setting. Using advanced imaging and molecular analysis, the researchers tracked how the cavity formed as cells reorganized.
Understanding how the blastocoel cavity forms offers critical insight into how early embryos organize themselves at the very start of life.
“Early development depends on precise coordination between molecular signals and physical forces,” said Professor Mo Li, who led the research at KAUST. “Our work uncovers the molecular machinery underlying this coordination, which drives the formation of one of the embryo’s earliest structures.”
The research forms part of KAUST’s Center of Excellence for Smart Health, which brings together expertise in stem cell biology, regenerative medicine and data-driven research to advance understanding of human health.
The research revealed how cells form a key structure in the earliest stage of human development:
In the first days after fertilization, a small cluster of cells reorganizes into a structure containing a fluid-filled cavity known as the blastocoel.
They identified a key driver of this process: a molecular pump known as V-ATPase. By moving charged particles within cells, the pump helps regulate how fluid is transported and accumulated between cells. As fluid builds, pressure increases, allowing the blastocoel cavity to form and expand.
The findings show how molecular activity within individual cells generates the physical forces required to form early embryo structures, providing a direct link between intracellular processes and the mechanics of embryo organization.
When the activity of the V-ATPase pump was disrupted, fluid could no longer accumulate, and the blastocoel structure failed to develop properly.
By improving the accuracy and reliability of human embryo models, this research provides a powerful new platform for studying these conditions and supporting research in reproductive medicine.