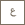New smart-drug research may help target cancer therapy

Dr. Niveen M. Khashab and her team at KAUST
Smart systems are a promising way to control the release of drugs within the body in order to produce enhanced and more targeted treatments. In a paper recently published in Biomaterials Science, (doi: 10.1039/c3bm60222b), Dr. Niveen M. Khashab, Assistant Professor of Chemical Science at KAUST, and colleagues, successfully demonstrated using thermosensitve liposomes to control the release of a drug by turning a simple system into a smart system.
“In this project we designed liposomes that are thermosensitive. By thermosensitive, I mean we used specific functional groups within the molecule that can get inside the surface of the liposomes and cause the drug to be released,” Dr. Khashab said.
Finding a Way to Target Drugs in the Body
Liposomes are highly used in drug delivery, but the problem is that they can’t be controlled or targeted. They are also quickly eliminated from the blood and often end up in the liver. Researchers have spent decades trying to reduce this problem. One of the ways is by studying stimuli-sensitive liposomes in hopes of finding an approach that would allow for a more controlled release– especially in cancer therapies.
“With cancer therapy and the current anti-tumor drugs in the market, a major disadvantage is that when the drugs are taken in the body, they can go everywhere. When trying to do a kind of smart-controlled release drug nano-vehicle, we are attempting to control the delivery,” Dr. Khashab said.
Liposomes Triggered by Heat to Control Drug Release
There are numerous approaches to stimuli-responsive liposomes ranging from liposomes that are pH sensitive, to magnetic fields, to experimenting with temperatures. Dr. Khashab and her team focused on the latter by designing thermosensitive liposomes that have proved to be effective in their release experiments inside cells.
Because the liposomes the team designed are heat sensitive, instead of releasing and spreading everywhere, the drug doesn’t release until it reaches the heated tumor tissue in the body. To do this, Dr. Khashab’s team used a cholesterol moiety that pins itself to the surface of the liposomes. She says using cholesterol as the “pin” was a logical choice as it makes up 30 percent of the cell membranes in the body, it is biocompatible and has a natural anchor-ability – something they needed to create what they call the “nail” or “comb” effect.
During their experiments, cholesterol modified NIPAm oligomers were used as the anchor that attached to the liposome. When they used a main-chain oligomer, it produced a “nail” effect by attaching to the surface head-to-head, and when they used a side-chain oligomer it produced a “comb” effect. Both effects proved to be efficient in the study.
“At normal body temperature, they would sit there on the surface. But if it is uptaken into a cancer cell and we increase the temperature to 40 degrees, you can monitor the ‘nail’ actually pressing inside,” Dr. Khashab said.
Rather than increasing the entire body temperature, the increase would be targeted to specific areas or tissues by external machines such as those used in hyperthermia and photodynamic therapy. As the tissues are heated, the cholesterol oligomer is aggregated, causing it to press into the lipid bilayer of the liposome. “This action of the pressing inside would release the drug inside the cancer cell essentially killing it. By this way we hope to have more control of drug release in cancer cells while keeping healthy cells safe,” Dr. Khashab explained.
The Difference Between the ‘Nail’ and the 'Comb’
The main difference between the “nail” and the “comb” release of the drug is the backbone shrinkage. With the “comb” effect, the side-chain sequence resulted in more lateral forces (the teeth of the comb) being pushed into the liposome as it was heated, which resulted in more of the drug being released at one time when compared to the “nail” effect.
Diagram showing the "nail" and "comb" effect.
However, their study showed that the difference in the results was not drastic due to the limited length of the chain. The team also found that controlling the length of the chain was another advantage of the NIPAm oligomers. The longer the chain; the faster the release.
Transforming a Simple System into a Smart System
“Fifty percent of the efforts in my lab go towards targeted and sustained drug delivery. What was nice about this project, was that we were able to take a simple system and modify it to be a smart system,” said Dr. Khashab.
Now that the team has shown the results of the their “nail” and “comb” effects on the release of liposomes in targeted areas, the next phase will be seeing how the effect works on live tissues and in various scenarios. “We were able to test and get effective results in-vitro, but the most critical step remains doing in-vivo work in small animals in order for this work to have major impact,” said Dr. Khashab.
By Michelle A. Ponto, KAUST News